Perthite
Two minerals
Life in the USA is not normal. It feels pointless and trivial to be talking about small looks at the fascinating natural world when the country is being dismantled. But these posts will continue, as a statement of resistance. I hope you continue to enjoy and learn from them. Stand Up For Science!
Perthite isn’t a mineral. It’s two minerals, usually a potassium feldspar (usually microcline or orthoclase, both K(AlSi3O8) but with different crystallography) and a plagioclase feldspar, usually albite Na(AlSi3O8). Although a solid solution, a homogeneous mixing in the chemistry from potassium (K) to sodium (Na), exists in the feldspars at higher temperatures, if the original K-Na aluminosilicate melt cools slowly, at lower temperatures a fully mixed K-Na feldspar can’t exist because of complexities in ion size and bond strengths. The two phases separate from each other to result in a complex intergrowth of potassium feldspar and albite as discrete zones called lamellae, thin layers that can be quite irregular. That intergrown texture is called perthite, from occurrences near the city of Perth, Ontario, Canada.
The process by which perthite forms is called exsolution. It is analogous to dissolution, but instead of going into solution, the two phases come out of (“ex-“) the solution that was homogeneous at higher temperature but which was not stable in a combined, single, solid phase at lower temperatures. Although in perthite it is usually just the two phases described above that form, some originally homogeneous solutions could exsolve into three or even more phases. It’s fair to think of “exsolution” as “unmixing.”
Perthite is probably the best-known example of exsolution, but it can occur in pyroxenes, sulfides, and other mineral pairs (or triples, or other combinations) that form homogeneous mixtures at higher temperatures. The resulting texture can range from quite uniform alternating layers to very ragged intergrowths to blob-like patches of one mineral within another. The intergrowths can be megascopic, as in my pieces, or only visible microscopically. And although in perthite the intergrowths are usually sub-parallel, exsolution can result in irregular, almost “wormy” textures as well.
Even though orthoclase and albite are different minerals, the way they intergrow during exsolution to form perthite is such that the resulting two-mineral mass works like one crystal, with the same (or almost the same) crystal faces and cleavage. Sometimes perthite lamellae even cross twin boundaries. Perthite is the word used to describe the result when there is more orthoclase or microcline (the host) than albite (the included lamellae). I think that is more common, but if the opposite situation occurs, the rock is by convention called antiperthite.
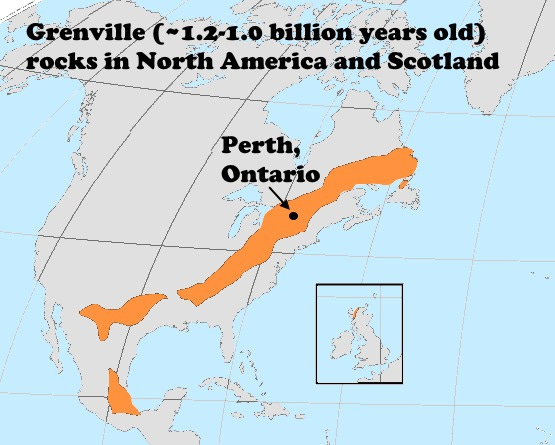
I collected the examples here from a road cut near Perth, Ontario, in August 1970, during the early part of a drive around the Gaspé Peninsula. The pink is the potassium feldspar, likely microcline based on analyzed specimens from the location, and the pink-white stringers are albite. In the photos you can see how intimately they are intergrown, almost in a feathery manner. The perthite comes from pegmatites that were emplaced between 1,250 million and 980 million years ago during the Grenville Orogeny, a collision between a relatively long, narrow continental band with the eastern and southern margin of Laurentia, the core of North America. The Grenville Orogeny made North America bigger.
Perth, Ontario, was named about 1816 to honor acting Governor-General Sir Gordon Drummond, whose ancestral home was Perthshire, Scotland, whose name is ultimately from a Pictish word for a wood or copse.
Although the perthite from Ontario was discovered in 1841 by physician James Wilson and described (with significant errors) in 1842 by chemist Thomas Thompson, it wasn’t until 1861 that it was recognized as an intergrowth of two distinct feldspar minerals by a student at the University of Munich, Dietoricus Gerhard. Gerhard wrote in Latin, and it was August Breithaupt’s nearly simultaneous and more widely disseminated report that led to consensus about the nature of perthite (Breithaupt had named both orthoclase and microcline).
For a nice history of all this, see this blog by Christopher P. Brett.



So perthite is the solution to solid solution?
What pyroxenes or sulfide can be perthitic? Is it visible, like in feldspar?